Got Plasma? COVID-19 Survivors Encouraged To Donate For Possible Treatment
The Food & Drug Administration put out information today regarding a possible treatment for the novel coronavirus COVID-19.
The FDA has been collaborating with experts across the board to create a "medical product" to treat COVID-19.
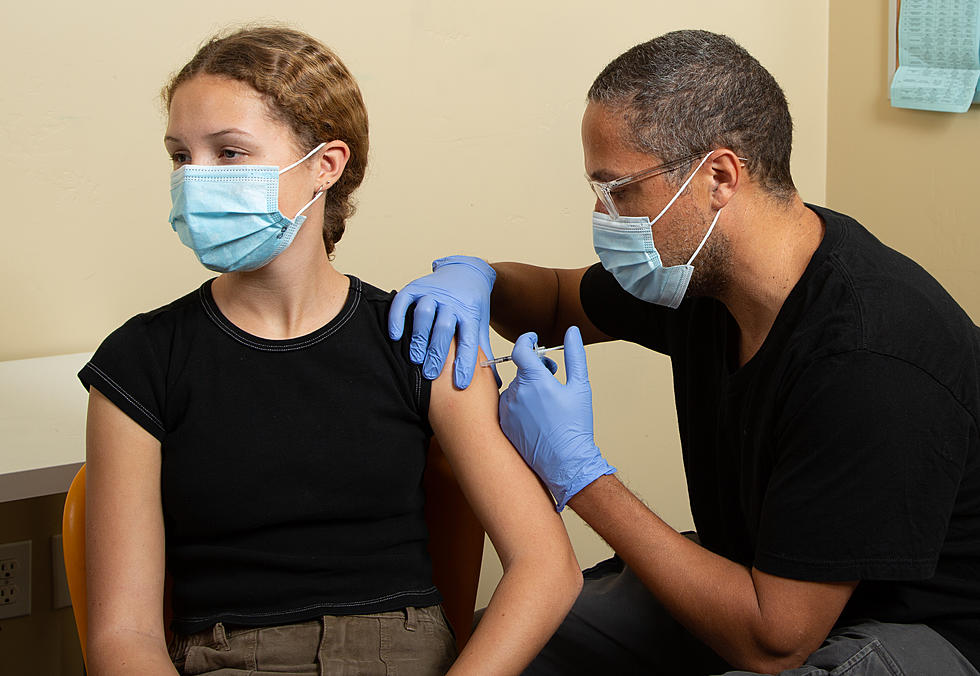
The information asks for the public's help, particularly those that have recovered from COVID-19. Information gathered from China suggests that plasma, a blood component that consists of lots of antibodies, could help lessen the severity of symptoms as well as shorten the length of time the illness may occur.
The FDA has created a webpage with information for those who have recovered and wish to donate their plasma for clinical trials. They are also working with the American Red Cross who can take donors as well.
Here is more information about the impact that one unit of plasma could have during the pandemic:
During this challenging time, many people are asking what they can do to contribute to the COVID-19 response. Those individuals who have recovered from COVID-19 could have an immediate impact in helping others who are severely ill. In fact, one donation has the potential to help up to four patients. Convalescent plasma can also be used to manufacture a biological product called hyperimmune globulin, which can similarly be used to treat patients with COVID-19.
The FDA is looking to recruit those that have fully recovered for at least two weeks. Contact your local blood or plasma collection center to schedule an appointment.
Places in Maine that do blood/plasma collection:
- American Red Cross Blood Center located in Bangor at 900B Hammond Street, call 1-800-RED-CROSS.
- American Red Cross Blood Center located in Auburn at Twin City Donor Center, 245 Center Street, call 1-800-RED-CROSS
- American Red Cross Blood Center located in Portland at 524 Forest Avenue, call 1-800-RED-CROSS
- BPL Plasma Donation Center located in Lewiston at 239 Main Street, call 207-783-3230
More From WBZN Old Town Maine